Acetone Polar or Nonpolar Solvent
Non-polar solvents are liquids or solvents that do not have a dipole moment. Is ethanol polar or nonpolar and why.

Is C3h6o Acetone Polar Or Non Polar Youtube
For example acetone C 3 H 6 O is an organic solvent but is polar as well.
. Water is a polar solvent. Polar solvents have a positive and a negative charge at different places in their structures and will dissolve other polar substances. Up to 24 cash back Is acetone polar or nonpolar solvent Acetone is an organic compound with its chemical formula CH32CO.
Acetone is a polar aprotic solvent. The bonds between the atoms have very different but measurable. It is classified as the simplest ketone.
An example of a polar solvent is water while ethanol is more nonpolar. A solvent is polar if it has a dipole moment greater than 16 D and a dielectric constant greater than 5. Acetone is another molecular material with both polar and nonpolar characteristics.
Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances while other solvents can only dissolve one or the other. So acetone is more polar than say hexane benzene and chloroform but less polar than acetonitrile acetic acid water and a saturated ammonium. ScCO 2 is a promising green solvent.
With a moderate relative static permittivity dielectric constant of 62 it dissolves not only polar. Acetone Dicholoroethane Tetrahydrofuran Dicholoromethane Chloroform Diethylether Benzene Toluene Xylene Carbontetrachloride Cyclohexane Petroleum ether Hexane Pentane Non. Acetones chemical makeup includes.
Why is acetone a better solvent than water. NMR Chemical Shifts of Common Laboratory Solvents as Trace Impurities. Other polar solvents include.
Acetones chemical makeup includes. There are more and less polar solvents. Acetone is a relatively polar organic solvent.
The values in the table below except as noted have been extracted from. As a result of this the solvents are missing any partial positive or negative charges. Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances while other solvents can only dissolve one or the other.
Acetone has a dipole moment of 288 D and hence is a polar molecule despite possessing characteristics of both the polar and nonpolar substances. Acetone polar or nonpolar. Polar solvent is a type of solvent that has large partial charges or dipole moments.
In contrast in eg. See answer 1 Best Answer. Why is chloroform a non-polar.
What is solvent polarity. Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances while other solvents can only dissolve one or the other. Liquid acetic acid is a hydrophilic polar protic solvent similar to ethanol and water.
Here acetone has been added to water left tube and carbon tetrachloride right tube. It exists as a. The values for acetone.
Basically they have small. Not all organic solvents are nonpolar.

Polar Protic Polar Aprotic Nonpolar All About Solvents

Why Is Acetone A Good Solvent Properties Explanation Video Lesson Transcript Study Com
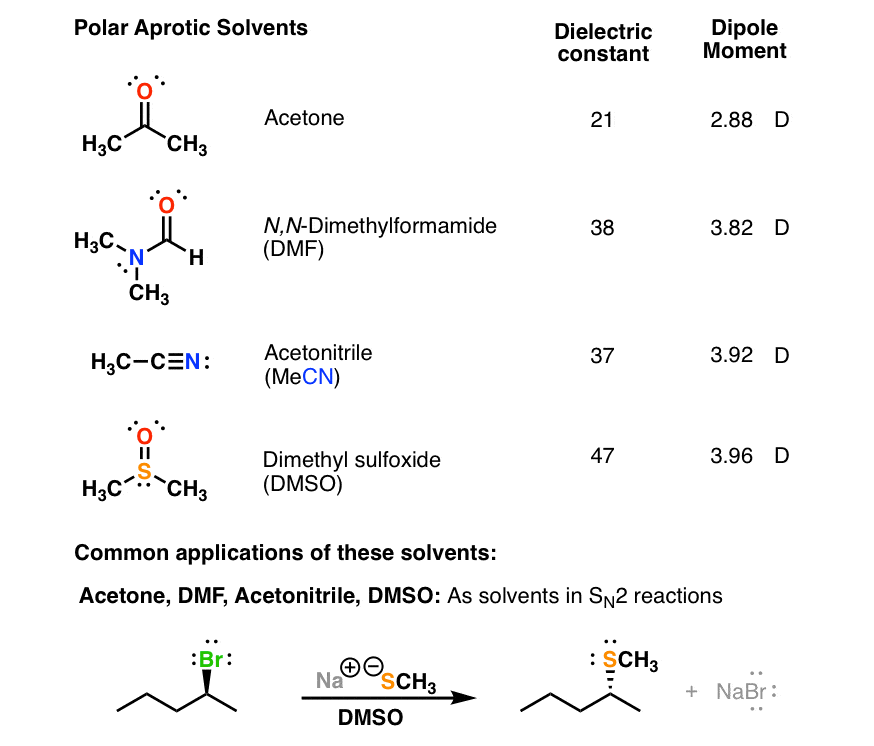
Polar Protic Polar Aprotic Nonpolar All About Solvents
Polar Vs Nonpolar Solvents Identifications And Examples Psiberg
No comments for "Acetone Polar or Nonpolar Solvent"
Post a Comment